In Chemical Solutions
There are a number of other environments where hot-dip galvanized steel is commonly specified for its longevity. Some environments where hot-dip galvanized steel can perform well are in chemical solutions. A primary factor governing galvanized coating corrosion behavior in liquid chemical environments is the solution's pH.
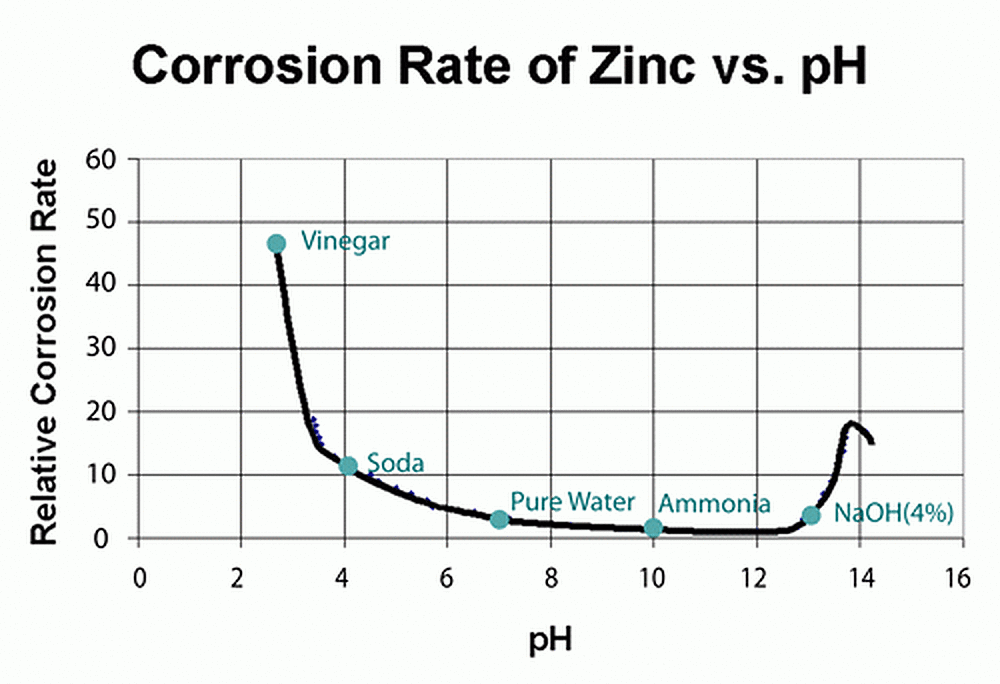
Galvanizing performs well in solutions of pH above 4.0 and below 12.5. This should not be considered a hard and fast rule because factors such as agitation, aeration, temperature, polarization, and the presence of inhibitors also may change the corrosion rate. Within the pH range of 4.0 to 12.5 a protective film forms on the zinc surface and the galvanized coating protects the steel by slowing corrosion to a very low rate. The protective film's exact chemical composition is somewhat dependent upon the specific chemical environment.
Since many liquids fall within the pH range of 4.0 to 12.5, galvanized steel containers are widely used for storing and transporting many chemical solutions. Corrosion occurs in countless solutions and chemical environments, but the actual corrosion rates will vary dramatically depending on the specifics of each setting. While zinc typically offers excellent protection from corrosion in most situations, there are occasionally certain conditions where chemicals can react with zinc to produce acidic products that will increase the corrosion rate.

Frank C. Porter, one of the foremost authorities on zinc, has provided a number of charts in his book Corrosion Resistance of Zinc and Zinc Alloys, which provide detailed information on how zinc reacts with a large variety of chemicals and solutions. Mr. Porter has been kind enough to allow us to share these charts with you. The charts are not for reproduction or distribution and provided for general information only. Caution should be used when reviewing the information because a slight difference in the specifics of your environment can lead to inaccurate conclusions (for example looking at the figures for a dry chemical powder versus a concentrated solution).
The chemicals are listed in a variety of fashions (i.e. solutions, vapors, dry, moist, immersed, with varying degrees of strengths, and so forth), so it is necessary to have a complete understanding of the setting in question before attempting to make any specific assumptions. Please contact us if you have any questions regarding the topic.
Corrosion Data for Zinc and Zinc Coatings with:
Inorganic Chemicals
Organic Chemicals
Detergents, Cleaners, Soaps, Synthetic Detergents
