To Certify or Not to Certify
A question that is often posed to the American Galvanizers Association (AGA) is why there isn’t an industry certification program for hot-dip galvanizers. The AGA and our members have long maintained the “certification” is in meeting the tenets in ASTM A123 Standard Specification for Zinc (Hot-Dip Galvanized) Coatings on Iron and Steel Products. However, in recent years, there has been more push and discussion about developing an independent certification program, particularly from the bridge and highway market. This is understandable considering the commonality of certification programs within the bridge market, and publically-funded projects. On the other hand, would a certification program actually accomplish the intended goal?
The reason behind the push for certification seems to be concern about inconsistent coating appearance and/or thickness. Unfortunately, there is very little the galvanizer can do within the process to control either of these factors. Coating appearance and thickness are most impacted by steel chemistry and fabrication techniques. However, the focus of a certification program for hot-dip galvanizing has been on process controls. The hard truth of the matter is implementing strict process controls will have little to no effect on these two biggest concerns from the marketplace. Furthermore, it is important to understand differences in coating appearance, although not always aesthetically pleasing, do not mean there is an issue with the galvanized coating quality. Matte gray and shiny finishes both provide long-term corrosion protection, which is what the hot-dip galvanized coating is intended to do.

The following information is intended to help specifiers further understand the variables that impact the final appearance of hot-dip galvanized coatings, and what is and is not under the galvanizer’s control. All of these variables should be taken into consideration and thoroughly understood before proceeding with the development of any type of certification program.
Hot-Dip Galvanizing (HDG)
Hot-dip galvanizing (HDG) is the process of dipping fabricated steel into a kettle or vat containing molten zinc. While the steel is in the kettle, the iron in the steel metallurgically reacts with the molten zinc to form a tightly-bonded alloy coating that provides superior corrosion protection to steel.
The hot-dip galvanizing (HDG) process consists of three basic steps:
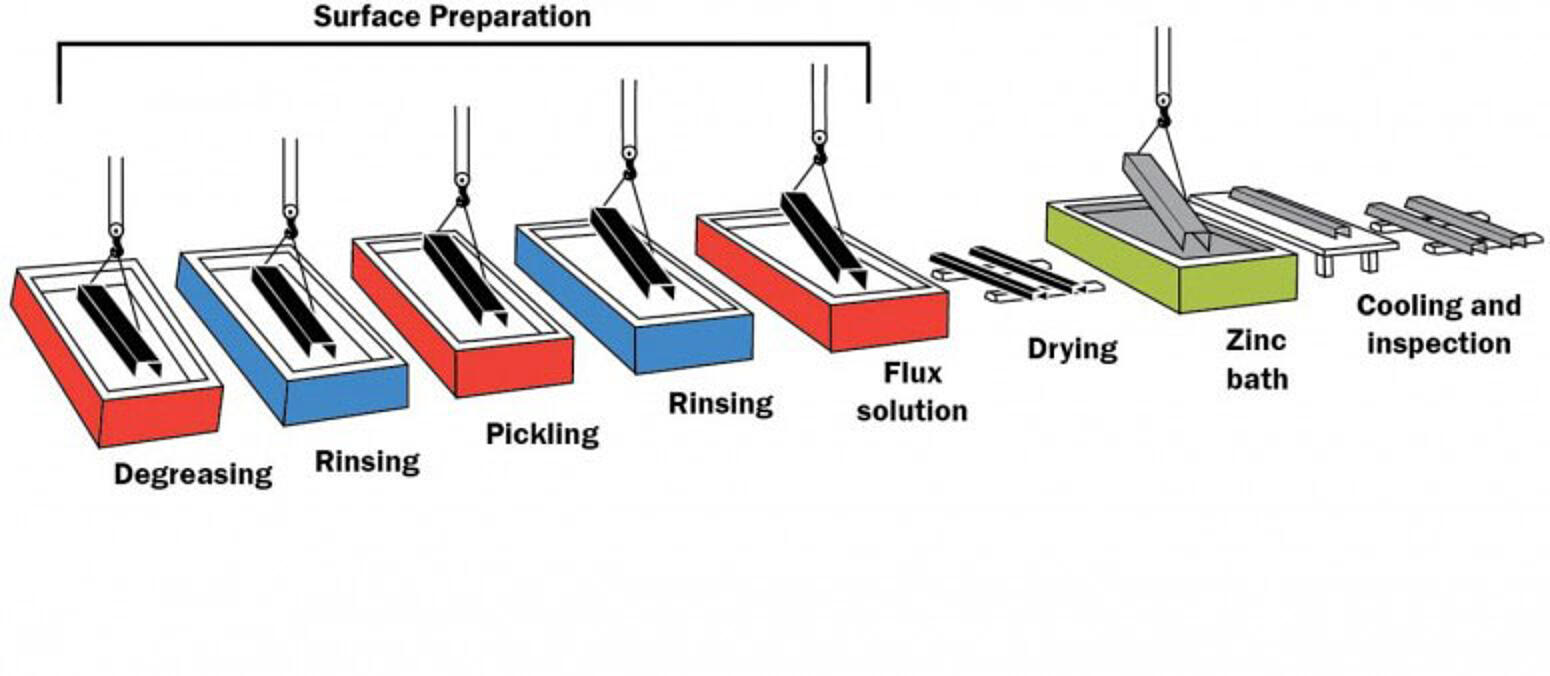
Surface Preparation
Surface preparation is a critical step in the application of any coating. The galvanizing process has its own built-in means of quality control because zinc will not react with an unclean steel surface. Any failures or inadequacies in surface preparation will be immediately apparent when the steel is withdrawn from the zinc bath because the unclean areas will remain uncoated, and immediate corrective action can be taken. Surface preparation for galvanizing consists of three steps:
Degreasing/Caustic Cleaning
A hot alkali solution, mild acidic bath, or biological cleaning bath removes organic contaminants such as dirt, paint markings, grease, and oil from the metal surface. Epoxy, vinyl, asphalt, or welding slag, which cannot be removed by degreasing, must be removed before galvanizing by grit-blasting, sand-blasting, or other mechanical means.
Pickling
A dilute solution of heated sulfuric acid or ambient hydrochloric acid removes mill scale and iron oxides (rust) from the steel surface. As an alternative to or in conjunction with pickling, this step can also be accomplished using abrasive cleaning or air blasting sand, metallic shot, or grit onto the steel. As long as the surface of the steel is completely cleaned to bare metal and all non-metallic substances removed, the galvanizing reaction is indifferent to the manner of cleaning.
Fluxing
The final surface preparation step in the galvanizing process, a zinc ammonium chloride solution, serves two purposes. It removes any remaining oxides from the surface of the steel, and deposits a protective layer on the steel to prevent any further oxides from forming on the surface prior to immersion in the molten zinc.
Galvanizing
During the actual galvanizing step of the process, the material is completely immersed in a bath of molten zinc. The bath chemistry is specified by ASTM A123 and requires at least 98% pure zinc by weight maintained at a temperature above the melting point of zinc. The zinc metal in the bath must meet the requirements of ASTM B6 or ASTM B960.
While the steel is immersed in the kettle, zinc reacts with iron in the steel to form a series of metallurgically-bonded zinc-iron intermetallic alloy layers, commonly topped by a layer of pure zinc metal. Once the fabricated items’ coating growth is complete, it is withdrawn slowly from the galvanizing bath, and the excess zinc is removed by draining, vibrating, and/or centrifuging. The metallurgical reaction will continue after the materials are withdrawn from the bath, as long as the steel part remains near bath temperature. Galvanized articles are cooled either by immersion in a passivation solution or water, or by being left in open air.
Inspection
The inspection of hot-dip galvanized steel is simple and quick. The two properties of the hot-dip galvanized coating most closely scrutinized are coating thickness and appearance/surface condition. A variety of simple physical tests can be performed to determine thickness, uniformity, adherence, and appearance.
Fabricator Controllable Variables
The following variables, which are under the fabricator’s control, can have a significant impact on the final HDG coating appearance and characteristics. To promote the best possible outcome from the hot-dip galvanizing process, the fabricator should understand:
- Steels with specific chemistry are known to result in galvanized coatings with thicknesses well above the minimum specified in ASTM A123.
-
ASTM A385 contains suggested chemistry for steels to be hot-dip galvanized. Steels with silicon and phosphorous outside the recommended ranges can result in excessively thick zinc coatings which can lead to delamination when impacted by outside forces during handling and erection. If the galvanizer is informed the steel is reactive, they can utilize bath additives and/or decrease the immersion time to try to limit the coating growth; however, this is not always effective or possible.
-
-
Proper design for vent and drain holes, cropped gusset plates, and seal-welded overlapped surfaces (prescribed in ASTM A385) will reduce or eliminate excess zinc buildup inside or outside the galvanized steel article.
-
Optimal immersion and removal speed from the zinc bath is a slow, steady rate. This allows even heating during immersion and even freezing of the zinc during removal. Too few or too small vent and drain holes, or improperly designed corners, make it difficult for the zinc to flow freely throughout the part leading to inefficiency and an uneven coating. Poor seal welds or stitch welding may cause ‘blowout’ of trapped cleaning chemicals from the overlapped surfaces leaving steel around the weld area ungalvanized.
-
-
Welds should be free of flux and slag, and created using weld rod similar in chemistry to the steel part.
-
Residual flux and slag are not always visible to the galvanizer, and the chemical cleaning steps in the galvanizing process will not remove them. The result is ungalvanized or loosely adherent coating areas around the weld. Similar to steel chemistry, certain elements in the weld rod chemistry can lead to more reaction with the zinc metal, which will likely yield a different appearance and coating thickness compared to the surrounding steel.
-
-
Stress-relieve steel or place a weld bead near the area of flame-cut cope edges.
-
The thermal gradient inherent in the galvanizing process affects the residual stresses in structural steel caused by flame-cut copes. If there is no stress-relieving prior to galvanizing, the cope cut areas may exhibit cracks after galvanizing. The fabricator may alternatively place weld bead on the beam web adjacent to the cope area, thereby relieving some of the residual stress in this area.
-
-
Minimize cold-working, but if necessary, do it in accordance with recommended bend radius, or incorporate stress-relieving methods.
-
Cold-working induces stress into structural steel and the excess residual stress can age the steel at galvanizing temperatures, thereby changing the steel properties. The heat of the galvanizing process accelerates the appearance of cracks (strain-age embrittlement), but does not cause them. The recommended practices for bend radius and stress-relieving are found in ASTM A143.
-
-
Use removable steel tags, weld bead, or stenciling to mark fabricated pieces.
-
Paint, lacquer, and some marking pen materials are not removed in the cleaning steps of the galvanizing process leaving uncoated areas. These marking must be mechanically removed prior to galvanizing.
-
Fabricator Uncontrollable Variables
In addition to the above mentioned variables within the fabricator’s control, there are a handful of variables in the fabrication process outside his/her control which can also result in varying galvanized coating appearance. Steel production is not a precise process in terms of uniform steel chemistry and distribution of elements throughout. Some fabrications require steels of varying chemistries and/or thicknesses to be combined, which influence the final coating appearance. The following implications should be understood:
- Fabrications using two or more steel thicknesses with the same steel chemistry
-
One of the advantages of hot-dip galvanizing is the total immersion of the steel part providing complete, uniform protection of all surfaces, tights joint areas, and internal spaces. When a fabrication comprised of two or more steel thicknesses is immersed, the thinner steel heats up to bath temperature sooner than the thicker steel. The reaction time between the iron in the steel and the zinc will be different and may result in two quite different appearances ranging from bright/shiny to matte gray.
-
-
Fabrications using steel with two or more different steel chemistries
-
The metallurgical reaction rate between the molten zinc and each of the steel types may be quite different and result in both a different coating thickness and different appearance, despite each part of the assembly being immersed in the same bath for the same amount of time. There is no way to modify the immersion time to produce a uniform thickness or appearance.
-
-
Fabrication volume insufficient to be galvanized in a compatible batch
-
To be as productive and efficient as possible, galvanizers will combine several jobs, which may contain quite different steel thicknesses and chemistries amongst its articles, and use wire to suspend the steel from a rack used to immerse all of the parts into the cleaning solutions and molten zinc at one time. There is no way to predict or ensure the appearance of the various steel pieces will be consistent. Even if all the steel articles were of the same thickness and an exact immersion time for the specific thickness was used, the coating thickness and appearance from piece to piece (and batch to batch) may be quite different.
-
-
Steel with a homogenous chemistry where all elements are evenly distributed throughout the steel article
-
Both the continuous cast and traditional iron ore/coke/limestone methods of producing steel do NOT guarantee the chemistry of the steel as reported on the mill test report is consistent throughout a beam, plate, or tube. In fact, it is normal for the chemistry from Point A to Point B on a structural steel piece to be slightly different. This difference in silicon and/or phosphorous, the two elements in steel that cause the most reactivity between the iron and the molten zinc, often results in a different galvanizing coating thickness and/or appearance on the same piece of steel. Even if the chemistry were precisely known and 100% homogenous throughout the piece of steel, having a prescribed immersion time and molten zinc temperature does not guarantee a predictable outcome. There are other unknown elements in the steel and possible rolled or fabricated stressed induced in the steel which may cause an area of matte finish adjacent to an area with a bright and shiny finish.
-
Galvanizer Variables
Extensive research and testing by the hot-dip galvanizing industry has led to the development of best practices (both in design and within the process) which are not 100% predictive of coating appearance and thickness, but deliver the most consistent quality within the parameters the process allows for the variety of materials being galvanized. In addition to the fabrication practices noted above, this is what we know about controls within the galvanizing process.
- The use of special high-grade, high-grade, and prime western zinc, produced from raw ore or via the re-melting of secondary zinc, does not guarantee a particular appearance or thickness.
- The addition of a small amount of nickel to the molten zinc bath helps to control the reactivity between iron and silicon as well as iron and phosphorous for some steel chemistries, but a specific range of coating thickness is not predictable for any thickness of steel.
- The addition of aluminum to the molten zinc bath generally results in a shinier zinc coating, but does not guarantee there will be no matte coating appearance on a part or all of a steel piece.
- There is no way to measure the degree of mill scale and corrosion on a piece of steel prior to immersion in the cleaning process tanks. Thus, the amount of time for the steel to residue in the acid bath is determined by experienced operators and is not available from a table of values.
- Pickling with hydrochloric or sulfuric acid are both effective means of removing mill scale and oxides. Over-pickling is possible when using sulfuric acid, but the resulting zinc coating is only marginally thicker than had the pickling time been exact. Galvanizers are careful to not over-pickle because applying excess zinc makes production less economical.
- Immediate water or passivation quenching after the steel is removed from the galvanizing bath stops the reaction between the iron and the zinc, usually resulting in a shinier coating, but not always. For tubular products, the quenching cools the material quickly and typically prevents peeling, i.e. the separation or pure zinc outer layer of the coating from the alloy layers. Based on symmetry and steel thickness, some fabrications cannot be quenched due to concerns related to distortion.
- Galvanizers regularly test and adjust the strength of their acid and flux solutions, and either completely change them out or employ a filtering system to maintain their effectiveness. Any variance from the optimal strength and/or cleanliness only affects the efficiency of the process and not the quality of the galvanized coating. If the flux fails, then this is immediately visible as ungalvanized areas on the fabrication upon removal from the zinc bath.
- Galvanizers regularly remove zinc-iron compounds called dross from the bottom of the galvanizing kettle where they settle over time. Effective “drossing” is important to maintain the kettle in proper working order and also minimizes the amount of dross that could potentially end up on the galvanized steel as it is removed from the kettle. The frequency of “drossing” the kettle is a function of how much steel volume is being processed and the cleanliness of the flux cleaning solution. This is an involved process and production cannot be done while “drossing;” thus, galvanizers are flexible on scheduling the drossing to ensure they meet customer demand.
- Prior to steel articles and fabrications being removed from total immersion in the zinc bath, galvanizers paddle zinc skimmings (zinc-oxide) to the side of the kettle so very little, if any, skimmings settle on top of the newly galvanized surface during removal from the bath. Because the oxidation of the molten zinc is a continuous process, it is not always possible to have all surfaces completely free of skimmings.
In summary, there are a number of variables within the production, design, and fabrication of steel to be galvanized that can have a profound effect on the final appearance of the HDG coating. These factors are outside the galvanizers’ control, and even if strict process controls are implemented within the hot-dip galvanizing process, as is common in certification programs, it does NOT guarantee more consistent coating appearances will be achieved. The concerns about inconsistency, though valid, should not be equated with inconsistent quality or inadequate protection. If the goal of a certification program is to achieve more consistent coating appearance and thickness, implementing controls within the galvanizing process will not warrant significant improvements. On the other hand, following the steel selection, fabrication and design best practices already outlined in ASTM A143, A384, and A385 more consistently will lead to more controllable galvanized coating appearances and thicknesses.

